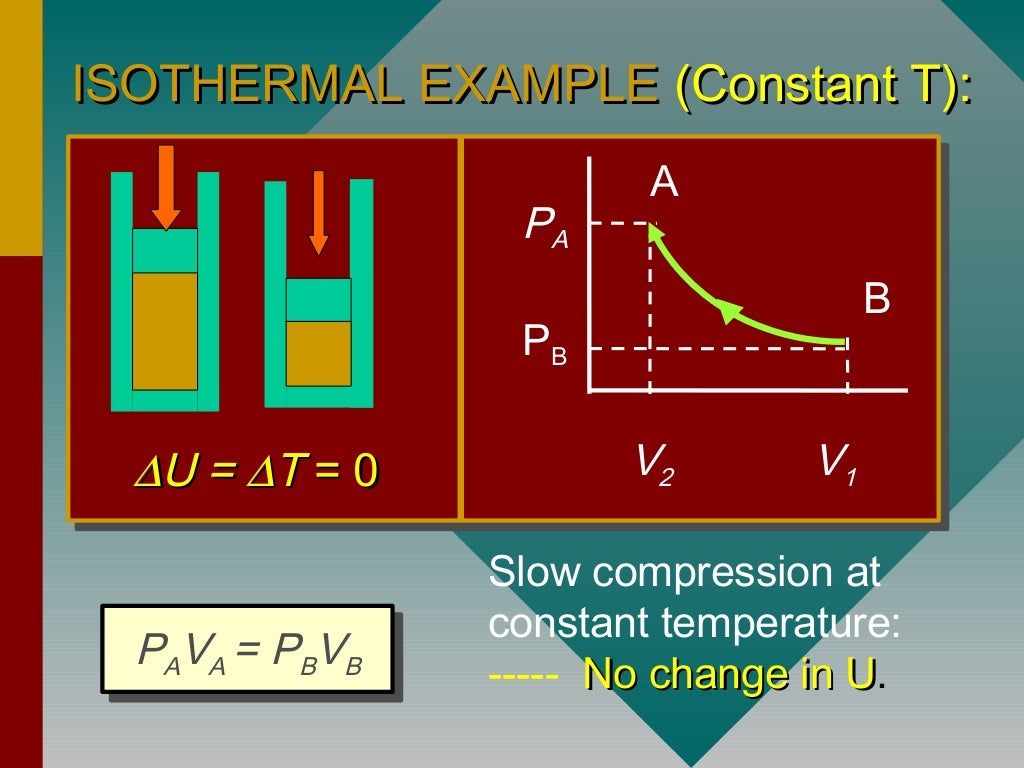
Although it isn't trivial in general, you can check how the formula simplifies for processes mentioned below. The general formula for work done by the gas is expressed as: ∫p(V)dV if we consider pressure as the function of volume. These parameters in real gases differ from theoretical ones, but we already contain them in our thermodynamic processes calculator. Note: We can also derive the expression for the work done by graphical method using a PV curve for the change in pressure and volume during an adiabatic expansion, then the area under the curve will give us the required work done. There is two specific heat in thermodynamic processes. If this box or system is thermally insulated, the process is known as free adiabatic expansion. There is no work done due to the absence of pressure.  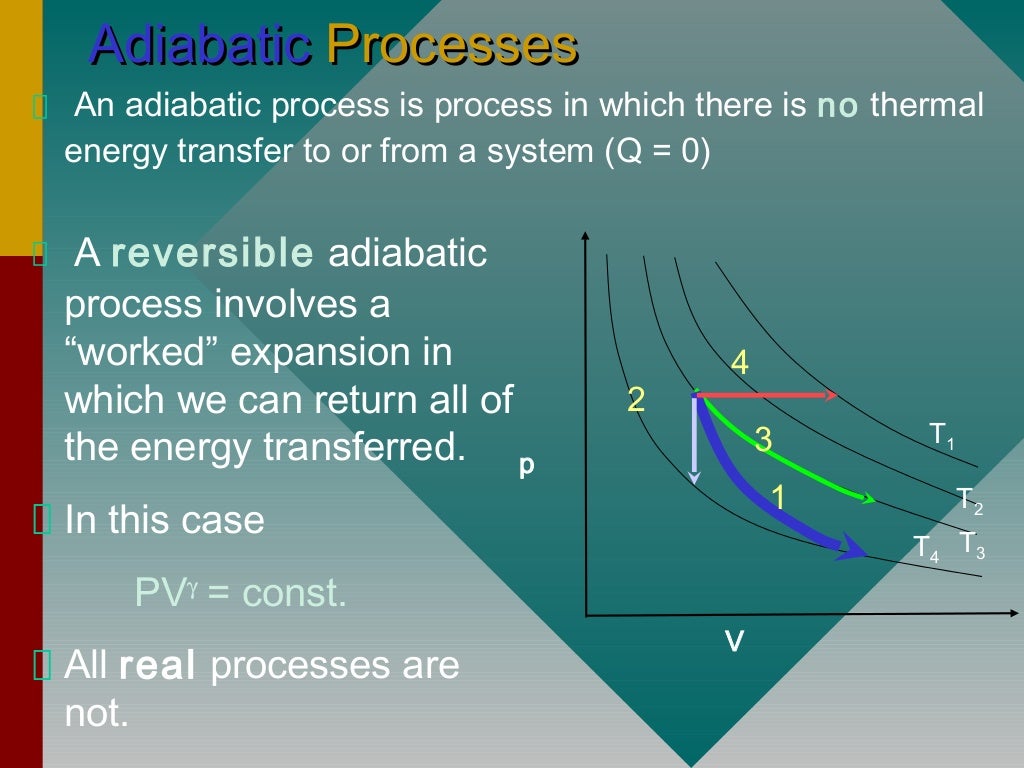
R for gases with more complex molecules. W R 1 T2 T1 This is the equation for work done in adiabatic expansion. This expansion process is caused due to volume, so pressure becomes zero.ΔT, where Cv is molar heat capacity under constant volume.Internal energy change is proportional to temperature variation ΔT and type of gas with the following equation: ΔU = Cv So the work is not transferred in the form of heat but change in volume. Since this process does not involve any heat. And since the volume is constant, so the work done is zero with dr cards. Or here we will have three times delta P. dW+dUdQ For adiabatic change dQ0 therefore dW-dU But dUCv dT therefore dW-Cv dT Cp-Cv R implies gamma Cv-CvR implies Cv(gamma-1) R implies Cv. So since the work done, so this works be delta V. Still, it is possible to find thermal energy changes ΔU, which are described by the first law of thermodynamics: ΔU = Q - W, where Q denotes heat absorbed, and W is work done by gas. Work done in reversible adiabatic process The heat transfer is zero in reversible adiabatic process. Because there is no external pressure for the gas to expand against, the work done by or on the system is zero. So until the pressure falls to so the gas again then heated and it provides the constant temperature. It's quite tricky to estimate the precise value of internal energy. Internal energy U is the sum of all kinds of energy present in a system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
